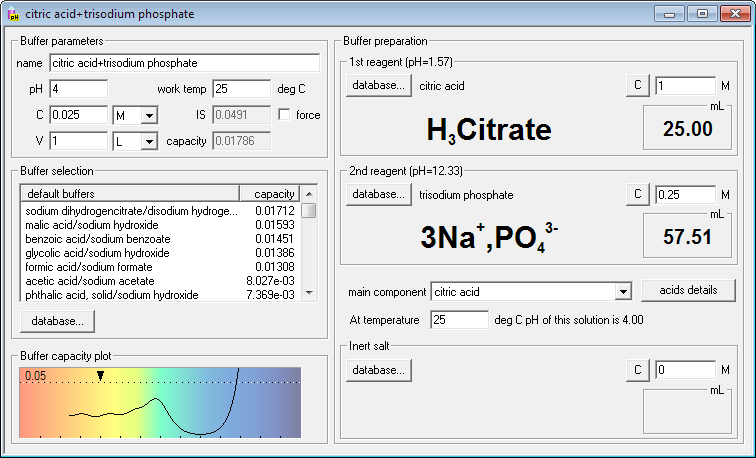
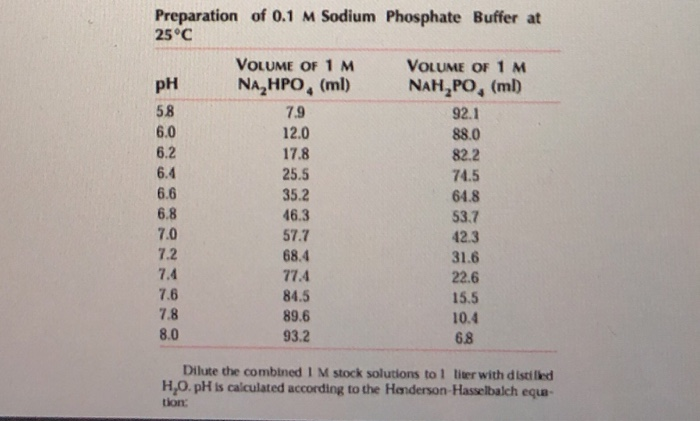
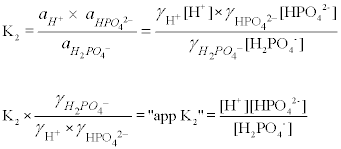SOLVED: Calculate the amount of monobasic potassium phosphate needed to prepare 100 mL ofa M solution Molecular formula: KHZPOa Molecular Mass: 136.09 glmol Calculate the amount of dibasic potassium phosphate needed to

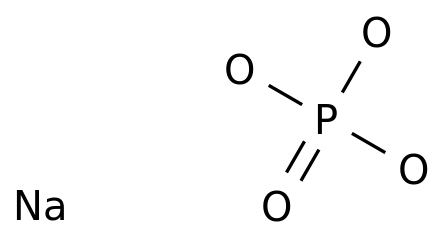
Sodium Phosphate Dibasic Heptahydrate (Na2HPO4.7H2O) Molecular Weight Calculation - Laboratory Notes

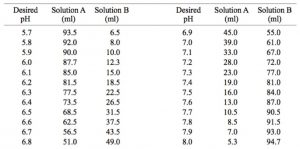
Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review