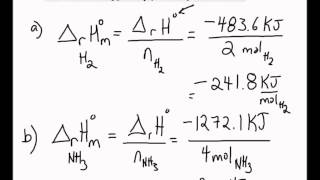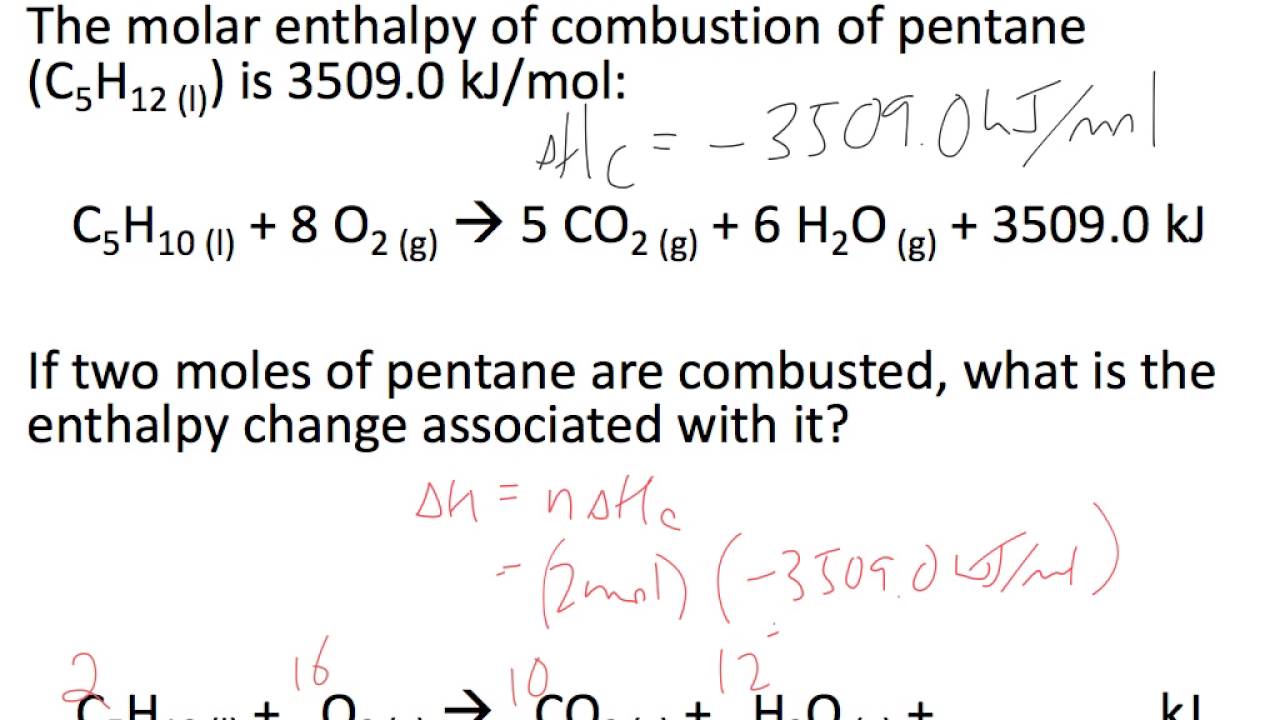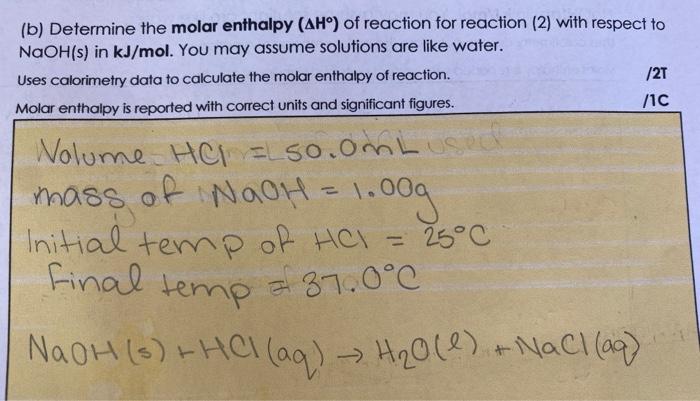Calculations in Chapter 10. Molar Enthalpy of Fusion Used when melting or freezing = ___energy ____ mol of substance Can be arranged to find any of the. - ppt download

3:04 calculate the molar enthalpy change (ΔH) from the heat energy change, Q - TutorMyself Chemistry
By the end of today's lesson you should · know what enthalpy is · · · understand the difference between a molar enthalpy v

Molar Enthalpies. use proper scientific terminology to describe molar enthalpies calculate molar enthalpies Calculate molar enthalpies using the. - ppt download

The molar enthalpy change for H2O(1) H2O(g) at 373 K and 1 atm is 41 kJ/mol. Assuming ideal behavior, the internal energy change for vaporization of 1 mol of water at 373