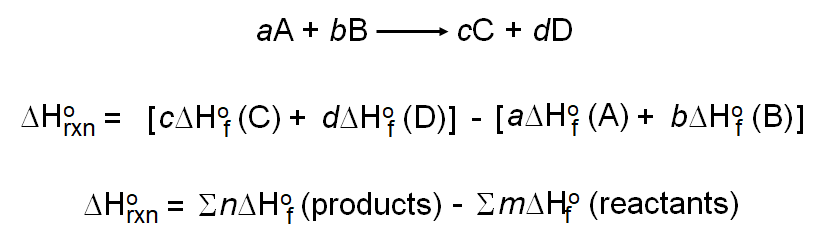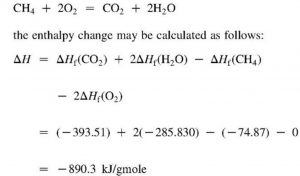Calculate the enthalpy change for the process CCl4(g)→ C(g) + 4Cl(g) and calculate bond enthalpy of C - Cl in CCl4(g) Δ vapH^ (CCl4) = 30.5 kJ mol ^-1 . Δ fH^ (

Calculate the enthalpy change where the standard heat of formation for gaseous NH(3) is -11.02 kcal/mol at 298 K. The reaction given is (1)/(2)N(2)(g)+(3)/(2)H(2)(g) rarr NH(3)(g)

Question Video: Determining the Standard Enthalpy of Formation of Ethanol Using Standard Enthalpies of Combustion | Nagwa